The moment I meet Horst Schmidt-Böcking outside the Bockenheimer Warte subway stop just north of the downtown area of Frankfurt, Germany, I know I have come to the right place. After my “Hi, thank you for meeting me,” his very first words are “I love Otto Stern.”
My trip on this prepandemic morning in November 2018 is to visit the place that, precisely a century before February 8, 2022, saw one of the most pivotal events for the nascent quantum physics. Without quite realizing what they were seeing, Stern and his fellow physicist and collaborator Walther Gerlach discovered quantum spin: an eternal rotational motion that is intrinsic to elementary particles and that, when measured, only comes in two possible versions—“up” or “down,” say, or “left” or “right”—with no other options in between.
Before the Roaring Twenties were over, physicists would reveal spin to be the key to understanding an endless range of everyday phenomena, from the structure of the periodic table to the fact that matter is stable—in other words, the fact that we don’t fall through our chair.
But the reason why I have a personal obsession with the Stern-Gerlach experiment—and why I am here in Frankfurt—is that it provided nothing less than a portal for accessing a hidden layer of reality. As physicist Wolfgang Pauli would explain in 1927, spin is quite unlike other physical concepts such as velocities or electric fields. Like those quantities, the spin of an electron is often portrayed as an arrow, but it is an arrow that does not exist in our three dimensions of space. Instead it is found in a 4-D mathematical entity called a Hilbert space.
Schmidt-Böcking—a semi-retired experimentalist at Goethe University Frankfurt and arguably the world’s foremost expert on Stern’s life and work—is the best guide I could have hoped for. We walk around the block from the station, past the Senckenberg Natural History Museum Frankfurt, to thePhysikalischer Verein, the local physicists’ society, which predates Goethe University Frankfurt’s 1914 founding. In this building, in the wee hours of February 8, 1922, Stern and Gerlachshot a beam of silver atoms through a magnetic field and saw that the beam neatly split into two.
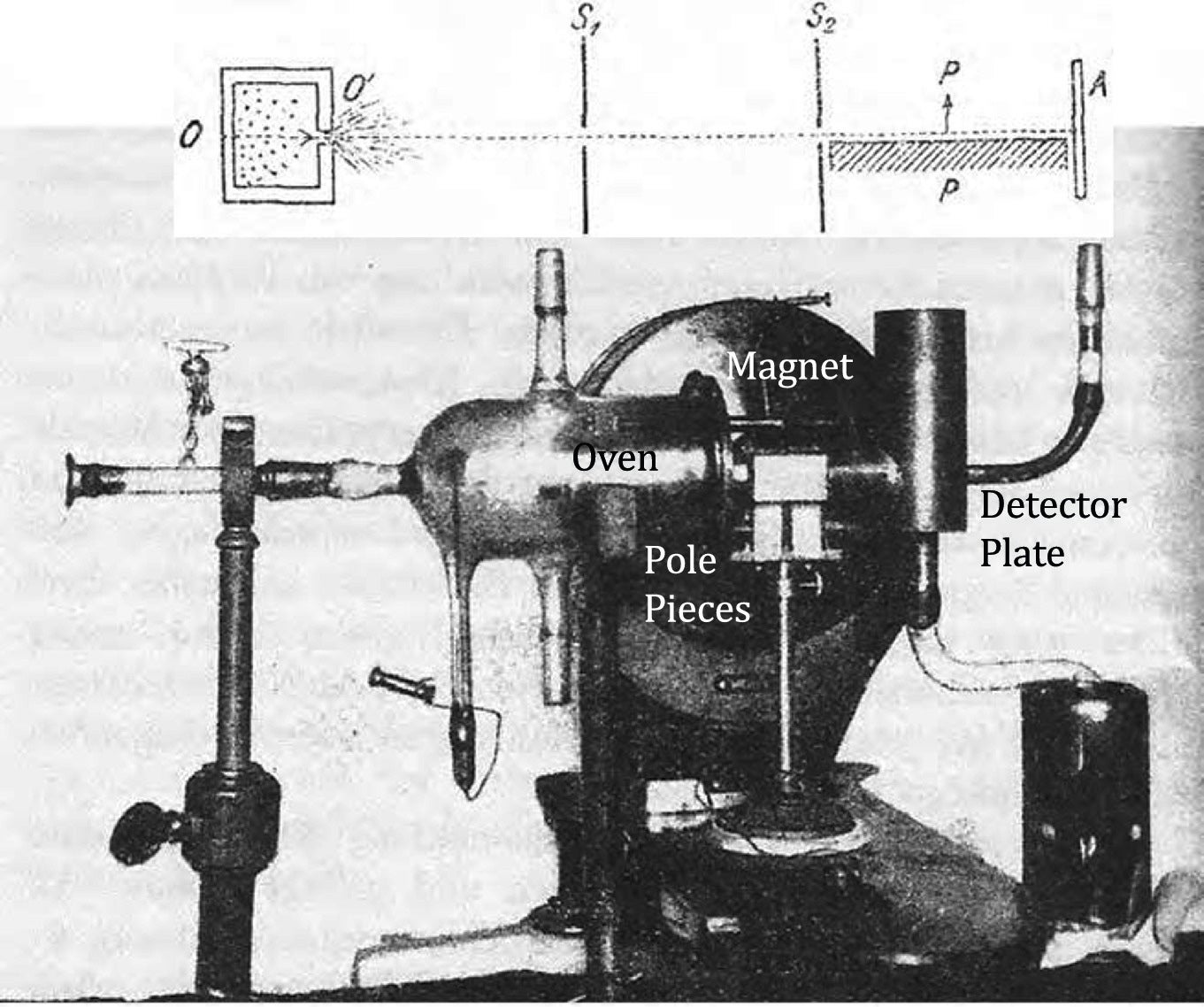
Once we are upstairs in the actual room of the experiment, Schmidt-Böcking explains that the whole experimental setup would have fit on a small desk. A vacuum system, made of custom blown-glass parts and sealed with Ramsay grease, enclosed the contraption. I find it hard to picture that in my mind, though, because the room, now windowless, is taken up by some of the nearby museum’s collections—specifically, cabinets with tiny specimens of bryozoans, invertebrates that form coral-like colonies.
Stern and Gerlach expected the silver atoms in their beam to act like tiny bar magnets and therefore to react to a magnetic field. As the beam shot horizontally, it squeezed through a narrow gap, with one pole of an electromagnet bracketed above and the other below. It exited the magnet and then hit a screen. When the magnetic field was turned off, the beam would just go straight and deposit a faint dot of silver on the screen, directly in line with the exit path of the beam from the magnet. But when the magnet was switched on, each passing atom experienced a vertical force that depended on the angle of its north-south axis. The force would be strongest upward if north pointed straight up, and it would be strongest downward if north pointed down. But the force could also take any value in between, including zero if the atom’s north-south axis was horizontal.
In these circumstances, a magnetic atom that came in at a random angle should have its trajectory deflected by a corresponding random amount, varying along a continuum. As a result, the silver arriving at the screen should have painted a vertical line. At least, that was Stern and Gerlach’s “classical” expectation. But that’s not what happened.
Unlike classical magnets, the atoms were all deflected by the same amount, either upward or downward, thus splitting the beam into two discrete beams rather than spreading it across a vertical line. “When they did the experiment, they must have been shocked,” says Michael Peskin, a theoretical physicist at Stanford University. Like many physicists, Peskin practiced doing the Stern-Gerlach experiment with modern equipment in an undergraduate lab class. “It’s really the most amazing thing,” he recalls. “You turn on the magnet, and you see these two spots appearing.”
Later that day in 2018, I get to see some of the original paraphernalia with my own eyes. Schmidt-Böcking drives me north in Frankfurt to one of the university’s campuses, where he keeps the artifacts inside well-padded boxes in his office. The most impressive piece is a high-vacuum pump—a type invented only a few years before the experiment—that removed stray air molecules using a supersonic jet of heated mercury.
It all looks tremendously fragile, and it is: According to witnesses, when the pieces were used, someglass part or other broke virtually every day. Restarting the experiment then required making repairs and pumping the air out again, which took several days. Unlike in modern experiments, the displacement of the beams was tiny—about 0.2 millimeter—and had to be spotted with a microscope.
At the time, Stern was shocked at the outcome. He had conceived the experiment in 1919 as a challenge to what was then the leading hypothesis for the structure of the atom. Formulated by physicist Niels Bohr and others starting in 1913, it pictured electrons like little planets orbiting the atomic nucleus. Only certain orbits were allowed, and jumping between them seemed to provide an accurate explanation for the quanta of light seen in spectroscopic emissions at least for the simple case of hydrogen. Stern disliked quanta, and together with his friend Max von Laue, he had pledged that “if this nonsense of Bohr should in the end prove to be right, we will quit physics.”
To test Bohr’s theory, Stern had set about exploring one of its most bizarre predictions, which Bohr himself did not quite believe: that in a magnetic field, atomic orbits can only lie at particular angles. To pursue this experiment, Stern realized that he could look for a magnetic effect of the electron’s orbit. He reasoned that the outermost electron of a silver atom, which according to Bohr is orbiting the nucleus in a circle, is an electric charge in motion, and it should therefore produce magnetism.
In Stern and Gerlach’s experiment, the physicists detected the splitting of the beam, which they saw as confirmation of Bohr’s odd prediction: The atoms got deflected—implying that they were magnetic themselves—and they did so not over a continuum, as in the classical model, but into two separate beams.
It was only after modern quantum mechanics was founded, beginning in 1925, that physicists realized that the silver atom’s magnetism is produced not by the orbit of its outermost electron but by that electron’s intrinsic spin, which makes it act like a tiny bar magnet.Soon after he heard about of Stern and Gerlach’s results, Albert Einstein wrote to the Nobel Foundation to nominate them for a Nobel Prize. But the letter, which Schmidt-Böcking discovered in 2011, was apparently ignored because it nominated other researchers as well, against the foundation’s rules. Stern did not quit the field. Eventually he was one of the most Nobel-nominated physicists in history, and he did get his prize in 1943, while World War II was raging.
Stern’s prize did not honor his work with Gerlach, however. Instead it was awarded for another tour de force experiment in which Stern and a collaborator measured the magnetism of the proton in 1933—shortly before the Nazi regime drove Stern out of Germany because of his Jewish background. That result was the earliest indication that the proton is not an elementary particle: we now know that it is made of three building blocks called quarks. Gerlach never won a Nobel Prize, perhaps because of his participation in the Nazi regime’s attempt to build an atomic bomb.
Today the concept of quantum spin as a 4-D entity is the foundation for all quantum computers. The quantum version of a computer bit, called the qubit, has the same mathematical form as the spin of an electron—whether or not it is in fact encoded in any spinning object. It often is not.
Even so, to this day, physicists continue to argue about how to interpret the experiment. According to now textbook quantum theory, initially, the silver atom’s outer electron does not know which way it is spinning. Instead it starts out in a “quantum superposition” of both states—as if its spin were up and down at the same time. The electron does not decide which way it is spinning—and therefore which of the two beams its atom travels in—even after it has skimmed through the magnet. When it has left the magnet and is hurtling toward the screen, the atom splits into two different, coexisting personas, as if it were in two places at the same time: one moves in an upward trajectory, and the other heads downward. The electron only picks one state when its atom arrives at the screen: the atom’s position can only be measured when it hits the screen toward the top or bottom—in one of the two spots but not both. Others take what they call a more “realist” approach: the electron knew all along where it was going, and the act of measurement is simply a sorting of the two states that happens at the magnet.
A recent prominent experiment seems to lend added credence to the former interpretation. It suggests that the two personas do coexist when the two spin states are separated. Physicist Ron Folman ofBen-Gurion University of the Negev in Israel and his colleagues re-created the Stern-Gerlach experiment using not individual atoms but a cloud of rubidium atoms. This was cooled to close to absolute zero, which made it act like a single quantum object with its own spin.
The researchers suspended the cloud in a vacuum with a device that can trap atoms and move them around using electric and magnetic fields. Initially, the cloud was in a superposition of spin up and spin down. The team then released it and let it fall by gravity. During its descent, they first applied a magnetic field to separate the atoms into two separate trajectories, according to their spin, just as in the Stern-Gerlach experiment. But unlike in the original experiment, Folman’s team then reversed the process and made the two clouds recombine into one. Their measurements showed that the cloud returned into its initial state. The experiment suggests that the separation was reversible and that quantum superposition persisted after being subject to a magnetic field that separated the two spin orientations.
The experiment goes to the heart of what constitutes a measurement in quantum mechanics. Were the spins in the Stern-Gerlach experiment “measured” by the initial sorting done by the magnet? Or did the measurement occur when the atoms hit the screen—or perhaps when the physicists looked at it? Folman’s work suggests that wherever a measurement happened, the separation was not at the first stage.
The results are unlikely to quell the philosophical diatribes around the meaning of quantum measurement, says David Kaiser, a physicist and historian of science at the Massachusetts Institute of Technology. But the impact of the Stern-Gerlach experiment remains immense. It led physicists to realize “that there was some internal characteristic of a quantum particle that really doesn’t map on to analogies to things like planets and stars,” Kaiser says.
